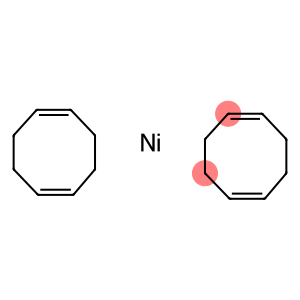
Nickel, bis((1,2,5,6-eta)-1,5-cyclooctadiene)-
Bis(1,5-cyclooctadiene)nickel(0)
CAS: 1295-35-8
Molecular Formula: C16H24Ni
Nickel, bis((1,2,5,6-eta)-1,5-cyclooctadiene)- - Names and Identifiers
| Name | Bis(1,5-cyclooctadiene)nickel(0) |
| Synonyms | CCRIS 5973 Bis(cyclooctadiene)nickel bis(1,5-cyclooctadiene)-nicke Bis-1,5-cyclooctadienylnickel Bis(1,5-cyclooctadiene)nickel Bis(1,5-cyclooctadiene)nickel(0) Nickel, bis(1,5-cyclooctadiene)- Bis((1,2,5,6-eta)-1,5-cyclooctadiene)nickel Nickel, bis((1,2,5,6-eta)-1,5-cyclooctadiene)- |
| CAS | 1295-35-8 |
| EINECS | 215-072-0 |
| InChI | InChI=1/2C8H12.Ni/c2*1-2-4-6-8-7-5-3-1;/h2*1-2,7-8H,3-6H2;/b2*2-1-,8-7- |
Nickel, bis((1,2,5,6-eta)-1,5-cyclooctadiene)- - Physico-chemical Properties
| Molecular Formula | C16H24Ni |
| Molar Mass | 275.06 |
| Melting Point | 60°C (dec.)(lit.) |
| Boling Point | 153.5°C at 760 mmHg |
| Flash Point | 31.7°C |
| Water Solubility | Soluble in benzene, toluene, terahydrofuran, ether, dimethyl formamide, hexamethylphosphoramide, N-methylpyrrolidinone. Insoluble in water. |
| Vapor Presure | 4.25mmHg at 25°C |
| Appearance | Bright gold crystalline powder |
| Color | Gold |
| Exposure Limit | NIOSH: IDLH 10 mg/m3; TWA 0.015 mg/m3 |
| Storage Condition | -20°C |
| Sensitive | Air Sensitive |
| MDL | MFCD00058902 |
| Use | Used to catalyze the cycloaddition reaction of 1, 3-diene compounds |
Nickel, bis((1,2,5,6-eta)-1,5-cyclooctadiene)- - Risk and Safety
| Risk Codes | R11 - Highly Flammable R40 - Limited evidence of a carcinogenic effect R45 - May cause cancer |
| Safety Description | S36/37 - Wear suitable protective clothing and gloves. S45 - In case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.) S16 - Keep away from sources of ignition. S53 - Avoid exposure - obtain special instructions before use. |
| UN IDs | UN 1325 4.1/PG 2 |
| WGK Germany | 3 |
| RTECS | QR6135000 |
| TSCA | No |
| HS Code | 29319090 |
| Hazard Class | 4.2 |
| Packing Group | I |
Nickel, bis((1,2,5,6-eta)-1,5-cyclooctadiene)- - Reference Information
| NIST chemical information | Information provided by: webbook.nist.gov (external link) |
| EPA chemical information | Information provided by: ofmpub.epa.gov (external link) |
| Application | Bis-(1, 5-cyclooctadiene) nickel is used as a catalyst for cycloaddition of 1, 3-diene compounds. It is used to catalyze the addition of allyl phenyl sulfide to alkynes to generate 1,4-diene compounds. |
| Use | Used to catalyze the cycloaddition reaction of 1, 3-diene compounds Used for the cycloaddition of 1, 3-diene compounds Catalyst. It is used to catalyze the addition of allyl phenyl sulfide to alkynes to generate 1,4-diene compounds. The reaction with terminal alkynes proceeds with high yield and high selectivity. Various functional groups can be connected. |
| Production method | 1. Under dry and oxygen-free conditions, in a 1-liter three-neck flask equipped with a three-way piston, a distillation head, a rubber cap and a heavy magnetic stirring rod, Place 57.0 grams of commercially available anhydrous bis (ethylphthalin acetone) nickel (1) and 500 milliliters of reagent pure toluene, toluene was distilled out in the presence of hydrogen to remove trace amounts of water remaining in the nickel salt. Replace the lower steamer head with a 250 ml constant pressure funnel and reduce the pressure to 0.01mm to remove all volatile substances and leave only 51.8 grams (0.201 moles) of bis (acetone) nickel (I) in the bottle. The system is vacuumized alternately (0.01mm) and filled with ammonia several times. In addition, toluene is distilled in another distillation device under the same conditions. After 50 ml of primary radium, 155 ml of toluene is collected with the above-mentioned constant pressure funnel and dropped into the above-mentioned flask containing double (acetone) nickel. Similarly, 123 ml [109g, 1.00 mol) of 1,5 cyclooctadiene is distilled into the constant pressure funnel and also dropped into the toluene solution of the above-mentioned bis (ethylphthaleacetone) nickel. Put a rubber cap on the funnel mouth, and then alternately vacuum and fill the system with ammonia several times to remove trace air. 5 grams (in weight gain) of butadiene are bubbled with a feeder. Into the reaction mixture. 113 ml of 25% (95.4g, 0.209 mol) triethylaluminum toluene solution was transferred into a constant pressure} I bucket by reduced pressure inhalation method, and then dropped into a cold but three-neck flask for 4 hours. When the original dark green solution becomes a brown-yellow rapid float in a short time, the reaction begins. After stirring at D ℃ for 5 hours, stirring at 25 ℃ for another 11 hours. Filter in inert gas to obtain yellow crystals. Dry in 0.01mm vacuum, weighing 38.4g 67%). Cold the filtrate to 78 ℃, and a small amount of product can be obtained. |
Last Update:2024-04-10 22:29:15
Supplier List
Featured Products
Product Name: Bis(1,5-cyclooctadiene)nickel(0), Crystal Request for quotationCAS: 1295-35-8
Tel: 15001012717
Email: biz5@infsci.com
Mobile: 15001012717
QQ: 3007471201
Product List: View Catalog
Spot supply
Product Name: Bis(1,5-cyclooctadiene)nickel Visit Supplier Webpage Request for quotationCAS: 1295-35-8
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Spot supply
Product Name: Bis(1,5-cyclooctadiene)nickel(0) Visit Supplier Webpage Request for quotationCAS: 1295-35-8
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Bis(1,5-cyclooctadiene)nickel(0) Request for quotation
CAS: 1295-35-8
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 1295-35-8
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: BIS(1,5-CYCLOOCTADIENE)NICKEL(0) Request for quotation
CAS: 1295-35-8
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 1295-35-8
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
Product Name: Bis(1,5-cyclooctadiene)nickel(0) Visit Supplier Webpage Request for quotation
CAS: 1295-35-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 1295-35-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Featured Products
Product Name: Bis(1,5-cyclooctadiene)nickel(0), Crystal Request for quotationCAS: 1295-35-8
Tel: 15001012717
Email: biz5@infsci.com
Mobile: 15001012717
QQ: 3007471201
Product List: View Catalog
Spot supply
Product Name: Bis(1,5-cyclooctadiene)nickel Visit Supplier Webpage Request for quotationCAS: 1295-35-8
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Spot supply
Product Name: Bis(1,5-cyclooctadiene)nickel(0) Visit Supplier Webpage Request for quotationCAS: 1295-35-8
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Bis(1,5-cyclooctadiene)nickel(0) Request for quotation
CAS: 1295-35-8
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 1295-35-8
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: BIS(1,5-CYCLOOCTADIENE)NICKEL(0) Request for quotation
CAS: 1295-35-8
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 1295-35-8
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
Product Name: Bis(1,5-cyclooctadiene)nickel(0) Visit Supplier Webpage Request for quotation
CAS: 1295-35-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 1295-35-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History





